Reducing Agents
Reducing agents are substances that lose electrons in redox reactions.?Generally speaking, the reactant with an increased valence of a substance contained is a reducing agent.?The reducing agent itself is reductive and oxidized, and its product is called oxidation product.?The reduction and oxidation reactions are carried out at the same time, that is to say, while the reducing agent carries out the redox reaction with the reduced object, it is also oxidized to become the oxidation product.
Typical reducing agents provided by Aladdin are as follows:
Active metal elements: such as Na, Al, Zn, Fe, etc
Active metal hydrides: such as lithium aluminum hydride LiAlH4
Some non-metallic elements: such as H2, C, Si, etc. elements (such as C, s, etc.)
Alkali metal elements: such as Li, Na, K, etc
Oxides at low valence: such as CO, SO2, etc.
Non metallic hydrides: such as H2S, NH3, HCl, CH4, etc.
Salts at low valence: such as Na2SO3, ferrous sulfate, FeSO4, etc.
Stannous chloride SnCl2, oxalic acid H2C2O4, potassium borohydride KBH4, sodium borohydride NaBH4, ethanol C2H5O
-
 Activity-Dependent Neuroprotective Protein Fragment 74-81, mouse, rat
Activity-Dependent Neuroprotective Protein Fragment 74-81, mouse, ratStarting at CN¥383.90
CAS : 211439-12-2
-

-
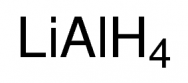
-
 Home
Home





 Hazardous Chemicals Business License (with storage)
Hazardous Chemicals Business License (with storage)