
基本描述
| 英文名称 | Trabectedin |
|---|---|
| 生化机理 |
Description: |
| 储存温度 | -20°C储存 |
| 运输条件 | 超低温冰袋运输 |
名称和标识符
| IUPAC Name | [(1R,2R,3R,11S,12S,14R,26R)-5,6',12-trihydroxy-6,7'-dimethoxy-7,21,30-trimethyl-27-oxospiro[17,19,28-trioxa-24-thia-13,30-diazaheptacyclo[12.9.6.13,11.02,13.04,9.015,23.016,20]triaconta-4(9),5,7,15,20,22-hexaene-26,1'-3,4-dihydro-2H-isoquinoline]-22-yl] acetate |
|---|---|
| INCHI | InChI=1S/C39H43N3O11S/c1-16-9-20-10-22-37(46)42-23-13-50-38(47)39(21-12-25(48-5)24(44)11-19(21)7-8-40-39)14-54-36(30(42)29(41(22)4)26(20)31(45)32(16)49-6)28-27(23)35-34(51-15-52-35)17(2)33(28)53-18(3)43/h9,11-12,22-23,29-30,36-37,40,44-46H,7-8,10,13-15H2,1-6H3/t22-,23-,29+,30+,36+,37-,39+/m0/s1 |
| InChi Key | PKVRCIRHQMSYJX-AIFWHQITSA-N |
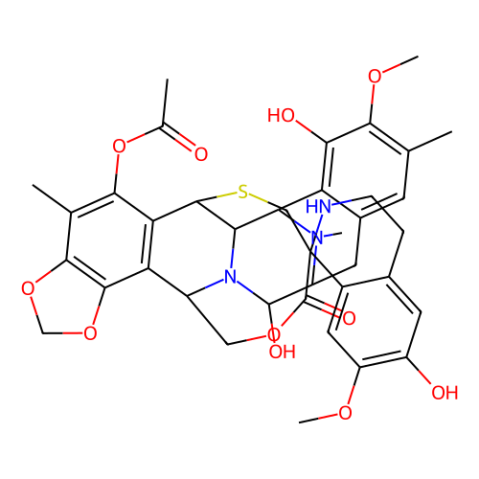
| Canonical SMILES | CC1=CC2=C(C3C4C5C6=C(C(=C7C(=C6C(N4C(C(C2)N3C)O)COC(=O)C8(CS5)C9=CC(=C(C=C9CCN8)O)OC)OCO7)C)OC(=O)C)C(=C1OC)O |
| 分子式 |
C39H43N3O11S |
| PubChem CID | 108150 |
| 分子量 | 761.84 |
数据库链接
| CAS Registry No. | 114899-77-3 |
|---|---|
| PubChem CID | 108150 |
| DrugBank Ligand | DB05109 |
| Wikipedia | Trabectedin |
| ChEMBL Ligand | CHEMBL450449 |
| ChEBI | CHEBI:84050 |
| RCSB PDB Ligand | ECT |
| DrugCentral Ligand | 4633 |
化学和物理性质
| 溶解性 | 25°C: DMSO |
|---|
安全信息
| 象形图 |

Toxic 
Health Hazard 
Harmful |
|---|---|
| 信号词 | Danger |
| 危险声明 |
H341: Suspected of causing genetic defects H373: Causes damage to organs through prolonged or repeated exposure H312: Harmful in contact with skin H332: Harmful if inhaled H300: Fatal if swallowed H361: Suspected of damaging fertility or the unborn child |
| 预防措施声明 | P261,P280,P302+P352,P321,P405,P501,P264,P260,P281,P271,P270,P304+P340,P362+P364,P330,P203,P301+P316,P318,P317,P319 |
靶标
| Target ID | 606 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 名称 | Pregnane X receptor | ||||||||||||||||||||||||
| 缩写名 | SHP2 | ||||||||||||||||||||||||
| 家族 | 1I. Vitamin D receptor-like receptors | ||||||||||||||||||||||||
| 别名 | STRL22 | ||||||||||||||||||||||||
| 基因和蛋白信息 |
|
||||||||||||||||||||||||
| Entrez Gene | 8856 (Hs) , 84385 (Rn) , 18171 (Mm) | ||||||||||||||||||||||||
| OMIM | 603065 (Hs) | ||||||||||||||||||||||||
| UniProtKB | O75469 (Hs) , Q9R1A7 (Rn) , O54915 (Mm) | ||||||||||||||||||||||||
| RefSeq Nucleotide | NM_033013 (Hs) , NM_052980 (Rn) , NM_010936 (Mm) , NM_001098404 (Mm) , NM_003889 (Hs) , NM_022002 (Hs) | ||||||||||||||||||||||||
| RefSeq Protein | NP_071285 (Hs) , NP_035066 (Mm) , NP_443212 (Rn) , NP_003880 (Hs) , NP_148934 (Hs) , NP_001091874 (Mm) | ||||||||||||||||||||||||
| Ensembl Gene | ENSG00000144852 (Hs) , ENSMUSG00000022809 (Mm) , ENSRNOG00000002906 (Rn) | ||||||||||||||||||||||||
| Protein GI | 16418481 (Rn) , 148536879 (Mm) , 34398348 (Hs) | ||||||||||||||||||||||||
| NURSA Receptor | 10.1621/IS7XZIEP9G (Hs) | ||||||||||||||||||||||||
| DrugBank Target | O75469 (Hs) | ||||||||||||||||||||||||
| CATH/Gene3D | 3.30.50.10 (N/A) | ||||||||||||||||||||||||
| ChEMBL Target | CHEMBL3401 (Hs) , CHEMBL2146315 (Rn) , CHEMBL1743244 (Mm) |
关联配体
| Ligand ID | 2774 |
|---|---|
| 名称 | trabectedin |
| 别名 | ET-743 |
| 类别 | Natural product |
| 学名 | 4-amino-N-(4,6-dimethylpyrimidin-2-yl)benzenesulfonamide |
| 生物活性评价 |
The sulfonamide class of antibacterial compounds are primarily bacteriostatic agents and have a broad spectrum of activity against both Gram-positive and Gram-negative species of bacteria (reviewed in |
| 评价 |
The ecteinascidins (Ets) are natural products derived from the marine tunicate Ecteinascidia turbinata. Being a natural substance, there is some ambiguity in the literature as to the exact chemical structure of trabectedin, therefore representations of the compound on the external resource links may vary slightly from that shown here. Trabectedin is now produced synthetically. Mechanistically trabectedin is a DNA minor groove binder and it forms covalent bonds with the exocyclic amino group of certain guanines in the minor groove and interstrand crosslinks via van der Waals interactions and hydrogen bonds |
参考文献
| 1. D'Incalci M, Galmarini CM. (2010) A review of trabectedin (ET-743): a unique mechanism of action.. Mol Cancer Ther, 9 (8): (2157-63). [PMID:20647340] |
| 2. Pommier Y, Kohlhagen G, Bailly C, Waring M, Mazumder A, Kohn KW. (1996) DNA sequence- and structure-selective alkylation of guanine N2 in the DNA minor groove by ecteinascidin 743, a potent antitumor compound from the Caribbean tunicate Ecteinascidia turbinata.. Biochemistry, 35 (41): (13303-9). [PMID:8873596] |
 首页
首页 400-620-6333
400-620-6333



 危险品化学品经营许可证(带存储)
危险品化学品经营许可证(带存储)