切换导航


搜索
基本描述
| 英文名称 | Alosetron |
|---|---|
| 生化机理 |
Alosetron is s Serotonin 5HT3-receptor antagonist that is used in treatment of irritable bowel syndrome. |
| 储存温度 | -20°C储存 |
| 运输条件 | 超低温冰袋运输 |
名称和标识符
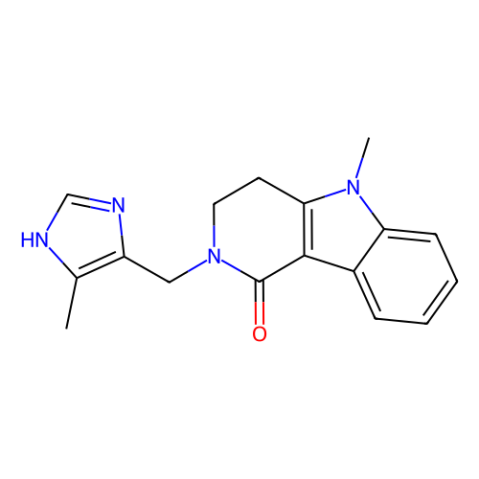
| IUPAC Name | 5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-3,4-dihydropyrido[4,3-b]indol-1-one |
|---|---|
| INCHI | InChI=1S/C17H18N4O/c1-11-13(19-10-18-11)9-21-8-7-15-16(17(21)22)12-5-3-4-6-14(12)20(15)2/h3-6,10H,7-9H2,1-2H3,(H,18,19) |
| InChi Key | JSWZEAMFRNKZNL-UHFFFAOYSA-N |
| Canonical SMILES | CC1=C(N=CN1)CN2CCC3=C(C2=O)C4=CC=CC=C4N3C |
| 分子式 |
C17H18N4O |
| PubChem CID | 2099 |
| 分子量 | 294.35 |
数据库链接
| PubChem CID | 2099 |
|---|---|
| DrugBank Ligand | DB00969 |
| CAS Registry No. | 122852-42-0 |
| ChEBI | CHEBI:253342 |
| ChEMBL Ligand | CHEMBL1110 |
| BindingDB Ligand | 50131429 |
| Wikipedia | Alosetron |
| RCSB PDB Ligand | S7Y |
| DrugCentral Ligand | 129 |
化学和物理性质
| 溶解性 | DMSO |
|---|
关联配体
| Ligand ID | 2296 |
|---|---|
| 名称 | alosetron |
| 别名 | GR68755 |
| 类别 | Synthetic organic |
| 学名 | 5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-3,4-dihydropyrido[4,3-b]indol-1-one |
| 生物活性评价 |
The sulfonamide class of antibacterial compounds are primarily bacteriostatic agents and have a broad spectrum of activity against both Gram-positive and Gram-negative species of bacteria (reviewed in |
| 评价 | Having been withdrawn by the manufacturer in 2000 following reports of severe adverse effects, the FDA approved a supplemental new drug application (2002) which allows remarketing of the drug, but under conditions of restricted use. |
 首页
首页 400-620-6333
400-620-6333



 危险品化学品经营许可证(带存储)
危险品化学品经营许可证(带存储)