
基本描述
| 规格或纯度 | Specific Activity >40 U/mg;Activity >5 U/ml |
|---|---|
| 英文名称 | Endo-H |
| 英文别名 | endo-beta-N-acetylglucosaminidase H |
| 来源 | recombinant gene from Streptomyces plicatus in E. Coli |
| 储存温度 | 2-8°C储存 |
| 运输条件 | 冰袋运输 |
| 产品介绍 |
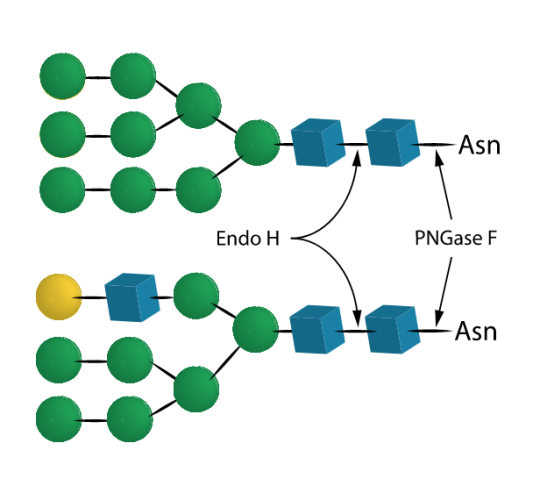
Product Description Endo H, endo-beta-N-acetylglucosaminidase H, Endoglycosidase H Endo H cleaves Asparagine-linked hybrid or high mannose oligosaccharides, but not complex oligosaccharides. It cleaves between the two N-acetylglucosamine residues in the diacetylchitobiose core of the oligosaccharide, generating a truncated sugar molecule with one N-acetylglucosamine residue remaining on the asparagine. In contrast, PNGase F removes the oligosaccharide intact. Detergent and heat denaturation may increase the rate of cleavage for some glycoproteins. Recommended Reagents included with: 1 vial: 5x Reaction Buffer - 400 ml 250 mM sodium phosphate, pH5.5 1 vial: Denaturation Solution - 200 ml 2% SDS, 1 M Beta-mercaptoethanol ? Molecular weight approximately 29 kD pH optimum: 5.5, active over the range 5-6. Applications Releases asparagine-linked hybrid or high mannose oligosaccharides, but not complex oligosaccharides. Endo H cleaves between the two N-acetylglucosamine residues in the diacetylchitobiose core of the oligosac-charide, generating a truncated sugar molecule with one N-acetylglucosamine residue remaining on the asparagine. In contrast, PNGase F removes the oligosaccharide intact. Detergent and heat denaturation may increase the rate of cleavage for some glycoproteins. Specific Activity One unit of Endo H activity is defined as the amount of enzyme required to catalyze the release of N-linked oligosaccharides from 1 μmole of denatured Ribonuclease B in one minute at 37?C, pH 5.5. Cleavage is monitored by SDS-PAGE (cleaved Ribonuclease B migrates faster).? Specificity Endo H cleaves Asparagine-linked hybrid or high mannose oligosaccharides, but not complex oligosaccharides. It cleaves between the two N-acetylglucosamine residues in the diacetylchitobiose core of the oligosaccharide, generating a truncated sugar molecule with one N-acetylglucosamine residue remaining on the asparagine. In contrast, PNGase F removes the oligosaccharide intact. Detergent and heat denaturation may increase the rate of cleavage for some glycoproteins. Formulation The enzyme is provided as a sterile-filtered solution in 20 mM Tris-HCl, 25mM NaCl, 1 mM EDTA (pH 7.5). Stability Several days exposure to ambient temperatures will not reduce activity. Stable at least 12 months when stored properly.? Quality & Purity Endo H is tested for contaminating protease as follows: 10 μg of denatured BSA is incubated at 37°C for 24 hours with 2 μl of enzyme. SDS-PAGE analysis of the treated BSA shows no evidence of degradation. The production host strain has been extensively tested and does not produce any detectable glycosidases. Directions for use 1. Add up to 200 μg of glycoprotein to an Eppendorf tube. Adjust to 37.5 μl final volume with de-ionized water. 2. Add 10 μl 5x Reaction Buffer 5.5 and 2.5 μl of Denaturation Solution. Heat at 100°C for 5 minutes. NOTE: It is not necessary to add Triton X-100. SDS will not inactivate Endo H. 3. Add 2.0 μl of Endo H to the reaction. Incubate 3 hours at 37°C. If SDS or heat denaturation is omitted, increase incubation time to at least 24 hours. Monitor cleavage by SDS-PAGE. The production host strain has been extensively tested and does not produce any detectable glycosidases. |
名称和标识符
| 酶学委员会编号 | 3.2.1.96 |
|---|
 首页
首页 400-620-6333
400-620-6333



 危险品化学品经营许可证(带存储)
危险品化学品经营许可证(带存储)