Buffer Reference Center
Buffer Reference Center
pH Ranges of Selected Biological Buffers Chart (25 °C, 0.1 M)
Tris or Trizma??Buffer Preparation – pH vs. Temperature
Phosphate Buffer Preparation – 0.2 M solution
Citric Acid – Na2HPO4?Buffer Preparation, pH 2.6-7.6
Citric Acid – Sodium Citrate Buffer Preparation, pH 3.0-6.2
Sodium Acetate – Acetic Acid Buffer Preparation, pH 3.7-5.6
Na2HPO4?– NaH22PO4?Buffer Preparation, pH 5.8-8.0 at 25 °C
Imidazole (glyoxaline) – HCl Buffer Preparation, pH 6.2-7.8 at 25 °C
Sodium Carbonate – Sodium Bicarbonate Buffer Preparation, pH 9.2-10.8
Buffer Preparation Formulas and Equations
CHOOSING THE RIGHT BIOLOGICAL BUFFER
Choose a buffer based on your pH requirements as well as the pKa, a measure of acid strength that accounts for pH, concentration, and temperature. Regulatory or purity needs for your exact application should also be considered. The following tables can help you navigate preparation of many common buffer solutions by pH and pKa.
Buffer Level
Buffer grade indicates the quality and impurity levels appropriate for different uses. We provide six grades of buffers indicated for general lab use, final pharmaceutical formulation and manufacturing, and applications in between that may need trace metal testing or materials of a specified purity.
USEFUL PH RANGES OF SELECTED BIOLOGICAL BUFFERS CHART (25 °C, 0.1 M)
|
TRIS BUFFER PREPARATION TABLE – PH VS. TEMPERATURE
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
?
PHOSPHATE BUFFER PREPARATION TABLE – 0.2 M SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
?
CITRIC ACID – NA2HPO4 BUFFER PREPARATION, PH 2.6–7.61?
Citric acid monohydrate, C6H8O7 ? H2O, MW 210.14; 0.1 M contains 21.01 g/L. Na2HPO4, MW 141.98; 0.2 M contains 28.40 g/L, or Na2HPO4 ? 2H2O, MW 178.05; 0.2 M contains 35.61 g/L.
x mL 0.1 M-citric acid and y mL 0.2 M-Na2HPO4?mixed
|
?
CITRIC ACID – SODIUM CITRATE BUFFER PREPARATION, PH 3.0-6.21
Citric acid monohydrate, C6H8O7?? H2O, MW 210.14; 0.1 M contains 21.01 g/L. Trisodium citrate dihydrate, C6H5O7Na3??2H2O, MW 294.12; 0.1 M contains 29.41 g/L.
x mL 0.1 M-citric acid and y mL 0.1 M-trisodium citrate mixed
|
SODIUM ACETATE – ACETIC ACID BUFFER PREPARATION, PH 3.7–5.61?
Sodium acetate trihydrate, CH3COONa ? 3H2O, MW 136.09; 0.2 M contains 27.22 g/L.
x mL 0.2 M-NaOAc and y mL 0.2 M-HOAc mixed
|
?
NA2HPO4?– NAH2PO4?BUFFER PREPARATION, PH 5.8-8.0 AT 25 °C1?
Na2HPO4? 2H2O, MW 178.05; 0.2 M contains 35.61 g/L. Na2HPO4 ? 12H2O, MW 358.22; 0.2 M contains 71.64 g/L. NaH2PO4 ? H2O, MW 138.01; 0.2 M contains 27.6 g/L. NaH2PO4 ? 2H2O, MW 156.03; 0.2 M contains 31.21 g/L.
x mL 0.2 M-Na2HPO4, y mL 0.2 M-NaH2PO4; diluted to 100 mL with H2O
|
?
IMIDAZOLE (GLYOXALINE) – HCL BUFFER PREPARATION, PH 6.2–7.8 AT 25 °C1
Imidazole,C3H4N2,, MW 68.08
25 mL 0.2 M-imidazole (13.62 g/L), x mL 0.2 M-HCl, diluted to 100 mL with H2O
|
SODIUM CARBONATE – SODIUM BICARBONATE BUFFER PREPARATION, PH 9.2-10.81
Na2CO3 ? 10H2O, MW 286.2; 0.1 M contains 28.62 g/L. NaHCO3, MW 84.0; 0.1 M contains 8.40 g/L.
x mL 0.1 M-Na2CO3and y mL 0.1 M-Na2HCO3mixed
|
BUFFER PREPARATION FORMULAS AND EQUATIONS
Percentage by weight (w/v)
(% buffer desired / 100) × final buffer volume (mL) = g of starting material needed.
Molar Solutions
desired molarity × formula weight × solution final volume (L) = grams needed
Henderson-Hasselbach Equation
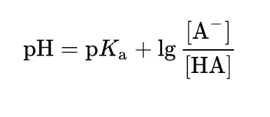
|
References
1. Dawson R, Elliot D, Elliot W, Jones KM. 1986. Data for Biochemical Research. 3rd ed.. Oxford Science Publ..
 首页
首页 400-620-6333
400-620-6333



 危险品化学品经营许可证(带存储)
危险品化学品经营许可证(带存储)